ARTICLE SUMMARY:
Abbott’s Aveir Conduction System Pacing leadless pacemaker system is in early feasibility trials. Excerpted from our recent feature article "Keeping the Beat".
Abbott is pushing ahead with a promising new approach to leadless cardiac pacing that relies on the heart’s natural conduction system to more closely mimic the heart’s own rhythm.
Abbott already markets the Aveir DR dual-chamber leadless pacemaker system, which includes the Aveir VR for the right ventricle and the Aveir AR for the right atrium, which can communicate with each other through Abbott’s i2i technology. Unlike traditional pacemakers, Aveir DR does not have wires—leads—connecting the pulse-generator to the heart or require a “pocket”; instead, the Aveir devices are fixed directly into the target area with a transfemoral catheter device. Leadless devices like Aveir DR avoid many of the risks of traditional transvenous leads, including lead fractures, dislodgement, and infection. (See “Bury the Lead: Abbott, Boston Scientific, and Medtronic Invest in Simpler CRM Devices,” MedTech Strategist, December 12, 2024.)
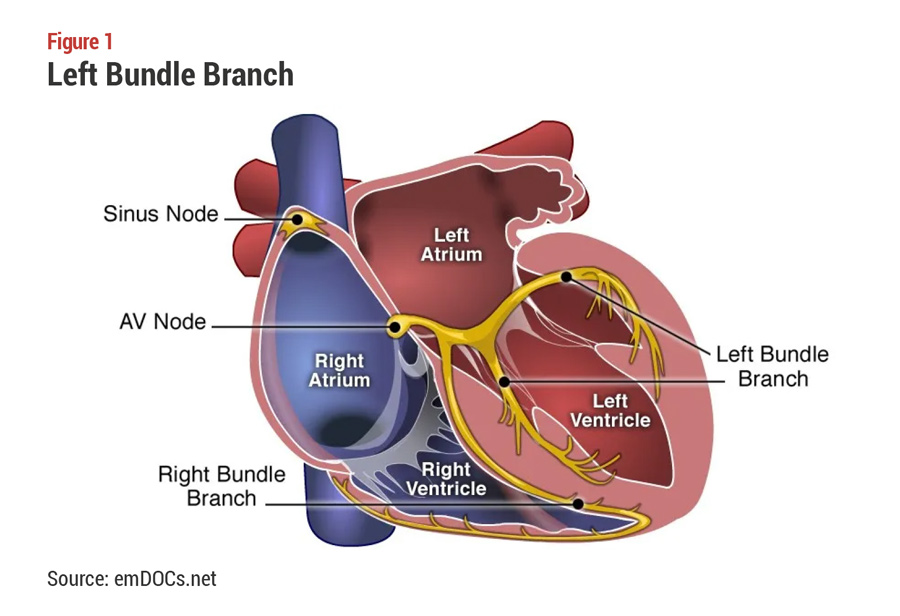 Because the Aveir system does not require a pocket and can be implanted wherever the catheter can reach, Abbott expects that leadless pacemakers will prove to be especially well-suited to directly pacing the left bundle branch area along the intraventricular septum (see Figure 1). From that position, the pacemaker can synchronize the contractions of both ventricles with the contractions of the atria.
Because the Aveir system does not require a pocket and can be implanted wherever the catheter can reach, Abbott expects that leadless pacemakers will prove to be especially well-suited to directly pacing the left bundle branch area along the intraventricular septum (see Figure 1). From that position, the pacemaker can synchronize the contractions of both ventricles with the contractions of the atria.
This technique is called conduction-system pacing or physiological pacing because it takes advantage of the heart’s natural electrical pathways. Five years ago, conduction-system pacing was rare, but now it represents around a third of pacemaker implants as it has the potential to overcome many of the challenges associated with traditional transvenous pacing approaches, Vish Charan, the product development leader in Abbott’s Cardiac Rhythm Management business, tells MedTech Strategist in an interview.
To bring the advantages of its leadless pacing technology to conduction-system pacing, Abbott is developing the Aveir Conduction System Pacing (CSP) leadless pacemaker system. Aveir CSP includes many of the features of the Aveir DR technology, but it is designed specifically for conduction-system pacing.
“We had to innovate significantly to go from the Aveir atrial device as we have it today, to the Aveir conduction-system pacing device, and we still have a lot more work to do,” Charan says. Aveir CSP has already been granted the FDA’s Breakthrough Device designation.
Abbott is sponsoring an early feasibility trial of Aveir CSP for pacing the left bundle branch area, which has been shown to be an optimal target for physiological pacing to restore electrical synchrony of the left ventricle. The feasibility study began late last year with procedures led by Petr Neužil, MD, PhD, at Na Homolce Hospital in Prague, Czech Republic, and Vivek Reddy, MD, at Mount Sinai Hospital in New York City. Reddy is the study's principal investigator.
The initial implants in the study represent the first-ever implants of a leadless pacemaker into the left bundle branch area, according to Abbott.
Abbott hopes the results of this initial study will be available in time for the Heart Rhythm 2025 conference in San Diego at the end of April. The company expects to follow the acute feasibility study with a chronic feasibility study that will evaluate Aveir CSP over a longer period and follow that with a chronic pivotal trial, but it has not announced a timeline for those trials.
